Search Result
Results for "
2-Deoxyuridine
" in MedChemExpress (MCE) Product Catalog:
2
Biochemical Assay Reagents
9
Isotope-Labeled Compounds
| Cat. No. |
Product Name |
Target |
Research Areas |
Chemical Structure |
-
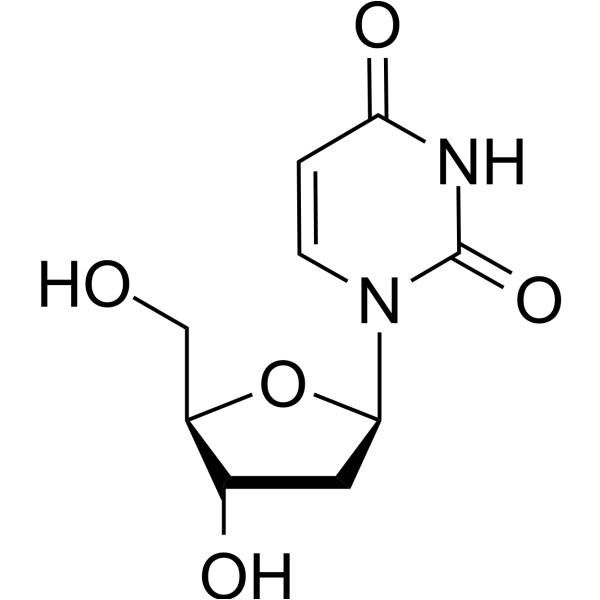
- HY-D0186
-
|
|
Endogenous Metabolite
|
Infection
|
|
2'-Deoxyuridine could increase chromosome breakage and results in a decreased thymidylate synthetase activity. A known use of 2'-Deoxyuridine is as a precursor in the synthesis of Edoxudine.
|
-
-
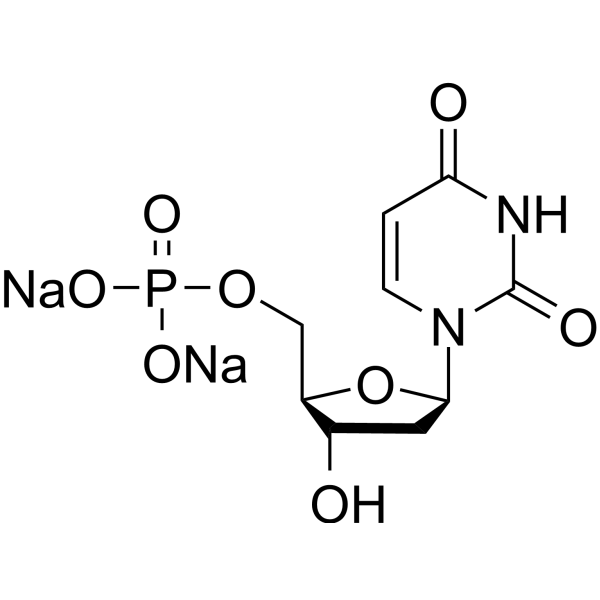
- HY-W011142
-
|
|
Endogenous Metabolite
|
Metabolic Disease
|
|
2'-Deoxyuridine 5'-monophosphate disodium is reductively methylated to dTMP (2'-deoxythymidine 5'-monophosphate) by bisubstrate enzyme thymidylate synthase (TS). dTMP is a nucleotide required for DNA synthesis .
|
-
-
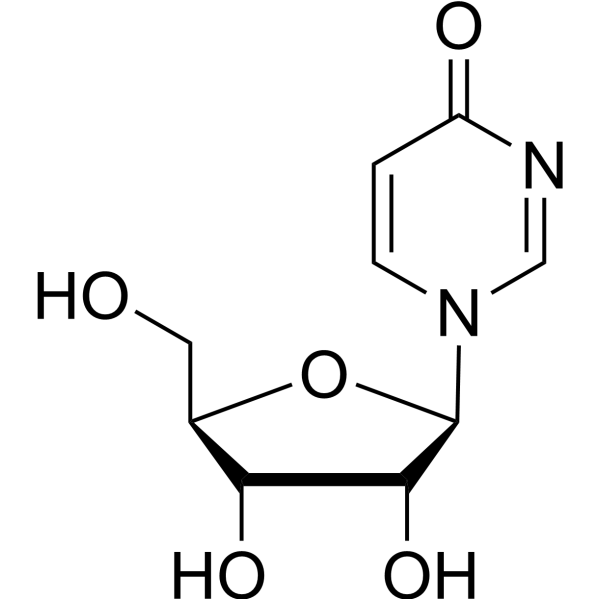
- HY-154329
-
|
|
Nucleoside Antimetabolite/Analog
|
Cancer
|
|
2-Deoxyuridine is a purine nucleoside analog. Purine nucleoside analogs have broad antitumor activity targeting indolent lymphoid malignancies. Anticancer mechanisms in this process rely on inhibition of DNA synthesis, induction of apoptosis, etc .
|
-
-
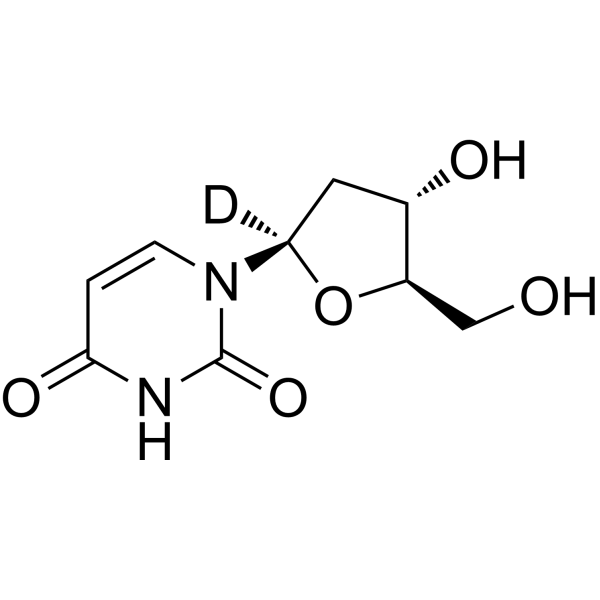
- HY-D0186S5
-
|
|
Isotope-Labeled Compounds
Endogenous Metabolite
|
Infection
|
|
2'-Deoxyuridine-d is the deuterium labeled 2'-Deoxyuridine. 2'-Deoxyuridine could increase chromosome breakage and results in a decreased thymidylate synthetase activity. A known use of 2'-Deoxyuridine is as a precursor in the synthesis of Edoxud[1]<
|
-
-
- HY-D0186S4
-
|
|
Isotope-Labeled Compounds
Endogenous Metabolite
|
Infection
|
|
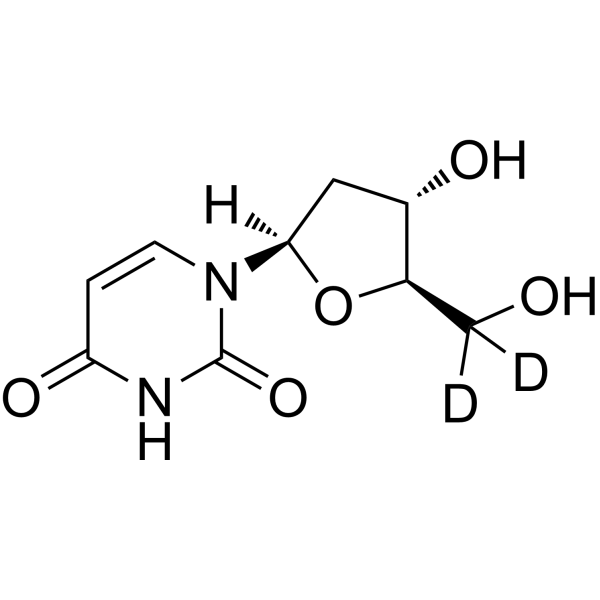
2'-Deoxyuridine-d2 is the deuterium labeled 2'-Deoxyuridine. 2'-Deoxyuridine could increase chromosome breakage and results in a decreased thymidylate synthetase activity. A known use of 2'-Deoxyuridine is as a precursor in the synthesis of Edoxud[1]
|
-
-
- HY-D0186S1
-
|
|
Isotope-Labeled Compounds
Endogenous Metabolite
|
Infection
|
|
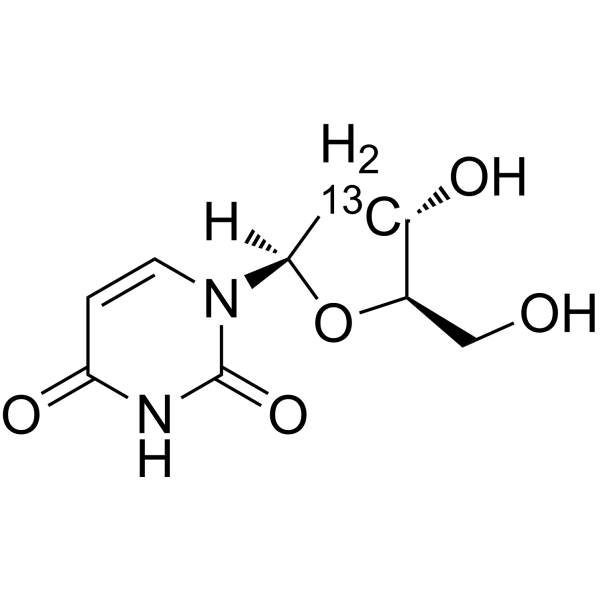
2'-Deoxyuridine-2′- 13C is the 13C labeled 2'-Deoxyuridine. 2'-Deoxyuridine could increase chromosome breakage and results in a decreased thymidylate synthetase activity. A known use of 2'-Deoxyuridine is as a precursor in the synthesis of Edoxud[1]
|
-
-
- HY-D0186S
-
|
|
Isotope-Labeled Compounds
Endogenous Metabolite
|
Infection
|
|
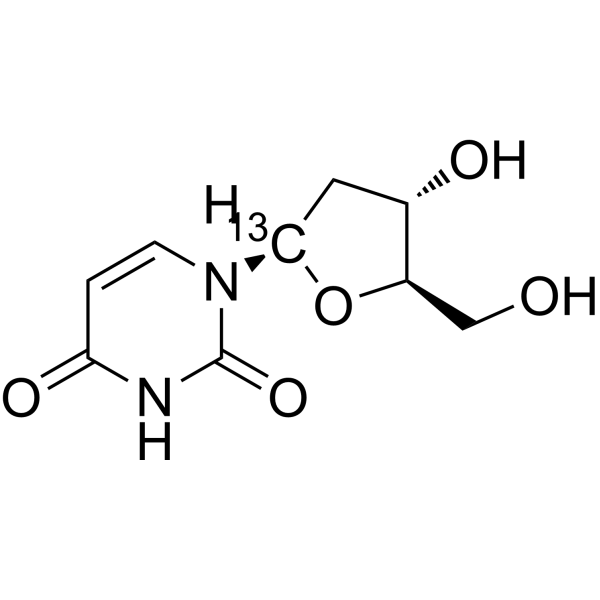
2'-Deoxyuridine-1′- 13C is the 13C labeled 2'-Deoxyuridine. 2'-Deoxyuridine could increase chromosome breakage and results in a decreased thymidylate synthetase activity. A known use of 2'-Deoxyuridine is as a precursor in the synthesis of Edoxud[1]
|
-
-
- HY-D0186S2
-
|
|
Isotope-Labeled Compounds
Endogenous Metabolite
|
Infection
|
|
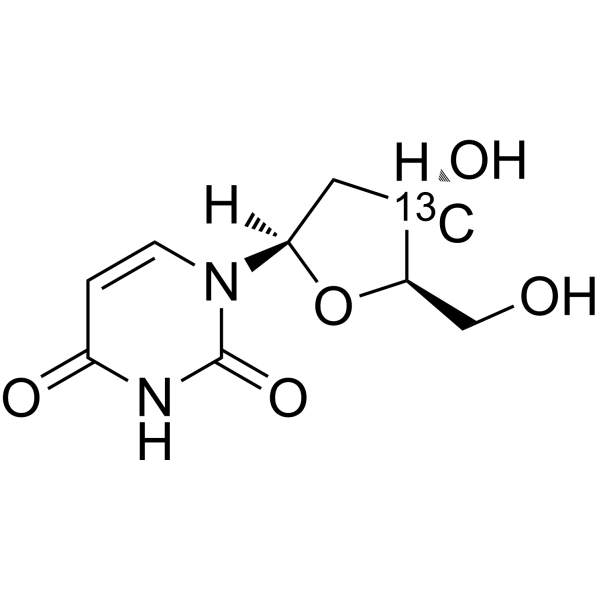
2'-Deoxyuridine-3′- 13C is the 13C labeled 2'-Deoxyuridine. 2'-Deoxyuridine could increase chromosome breakage and results in a decreased thymidylate synthetase activity. A known use of 2'-Deoxyuridine is as a precursor in the synthesis of Edoxud[1]
|
-
-
- HY-D0186S3
-
|
|
Isotope-Labeled Compounds
Endogenous Metabolite
|
Infection
|
|
2'-Deoxyuridine-5′- 13C is the 13C labeled 2'-Deoxyuridine. 2'-Deoxyuridine could increase chromosome breakage and results in a decreased thymidylate synthetase activity. A known use of 2'-Deoxyuridine is as a precursor in the synthesis of Edoxud[1]
|
-
-
- HY-D0186S8
-
|
|
Endogenous Metabolite
|
Infection
|
|
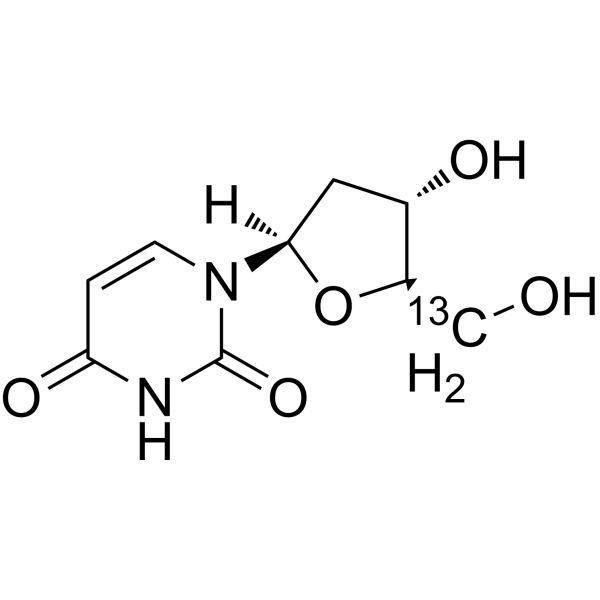
2'-Deoxyuridine-d2-1 is the deuterium labeled 2'-Deoxyuridine[1]. 2'-Deoxyuridine could increase chromosome breakage and results in a decreased thymidylate synthetase activity. A known use of 2'-Deoxyuridine is as a precursor in the synthesis of Edoxudine[2].
|
-
-
- HY-D0186S7
-
|
|
Endogenous Metabolite
|
Infection
|
|
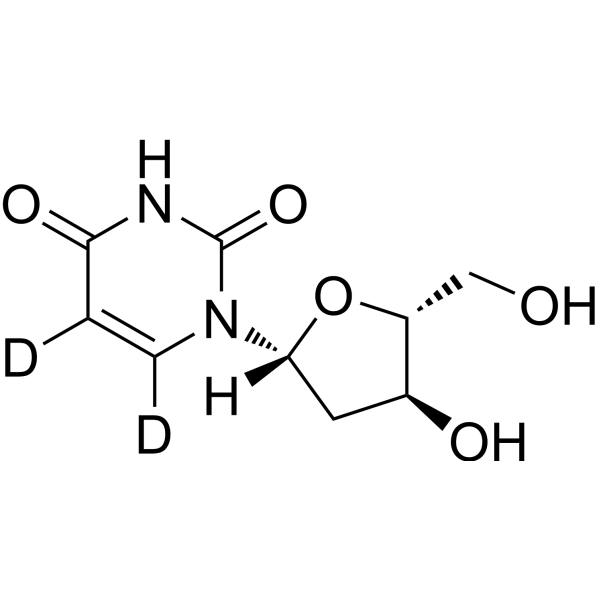
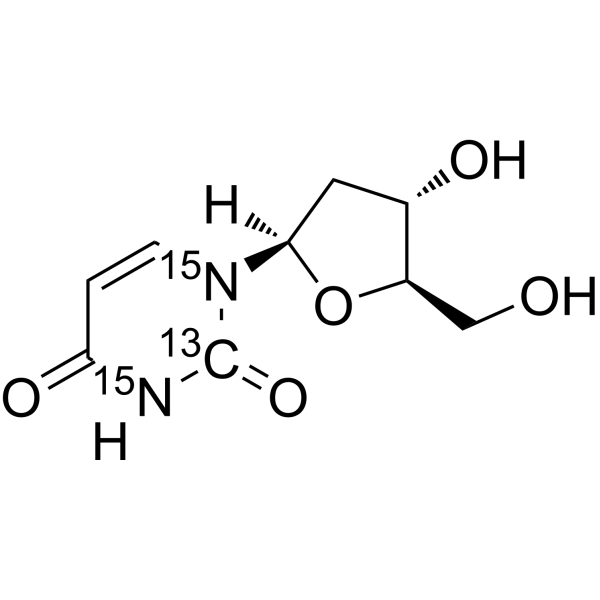
2'-Deoxyuridine- 13C, 15N2 is the 13C and 15N labeled 2'-Deoxyuridine[1]. 2'-Deoxyuridine could increase chromosome breakage and results in a decreased thymidylate synthetase activity. A known use of 2'-Deoxyuridine is as a precursor in the synthesis of Edoxudine[2].
|
-
-
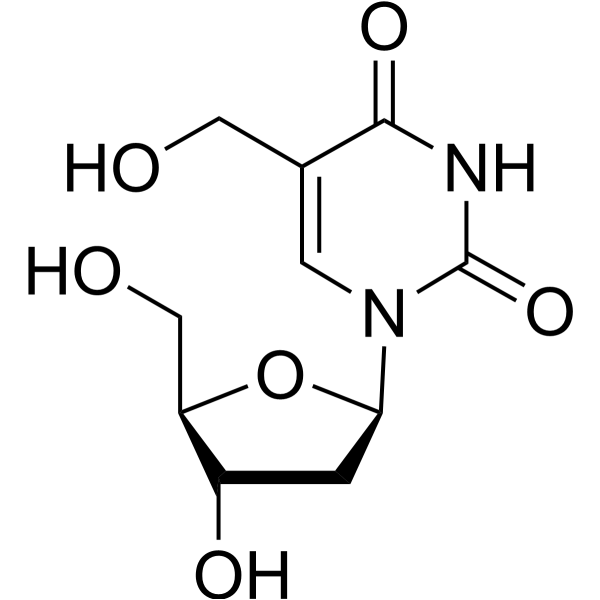
- HY-129983
-
|
|
HSV
|
Infection
Cancer
|
|
5-Hydroxymethyl-2'-deoxyuridine is a nucleoside analog. 5-Hydroxymethyl-2'-deoxyuridine inhibits the replication of multiple human leukemia cell lines with IC50 values of 1.7-5.8 μM. 5-Hydroxymethyl-2'-deoxyuridine prolongs the survival of mice carrying L1210 leukemia. 5-Hydroxymethyl-2'-deoxyuridine can be used for the research of cell replication and leukemia [2] .
|
-
-
- HY-W424779
-
|
|
Drug Metabolite
|
Infection
|
|
5-Carboxy-2′-deoxyuridine is a metabolite of Trifluridine . 5-Carboxy-2′-deoxyuridine is a methyl oxidation product of Thymidine that can be formed by menadione-mediated photosensitization of Thymidine [2].
|
-
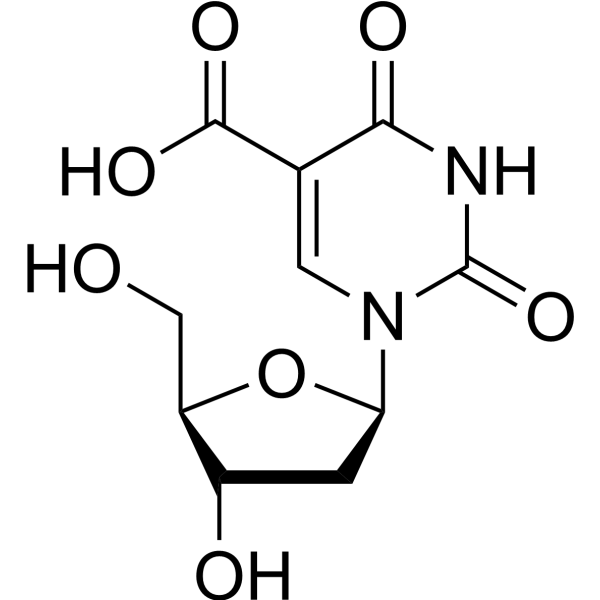
-
- HY-118411
-
|
|
PROTAC Linkers
|
Cancer
|
|
5-Ethynyl-2'-deoxyuridine (EdU), a thymidine analogue, is incorporated into cellular DNA during DNA replication and the subsequent reaction of EdU with a fluorescent azide in a “Click” reaction. EdU staining is a fast, sensitive and reproducible method to study cell proliferation . 5-Ethynyl-2'-deoxyuridine is an alkyl chain-based PROTAC linker that can be used in the synthesis of PROTACs [2]. 5-Ethynyl-2'-deoxyuridine is a click chemistry reagent, it contains an Alkyne group and can undergo copper-catalyzed azide-alkyne cycloaddition (CuAAc) with molecules containing Azide groups.
|
-
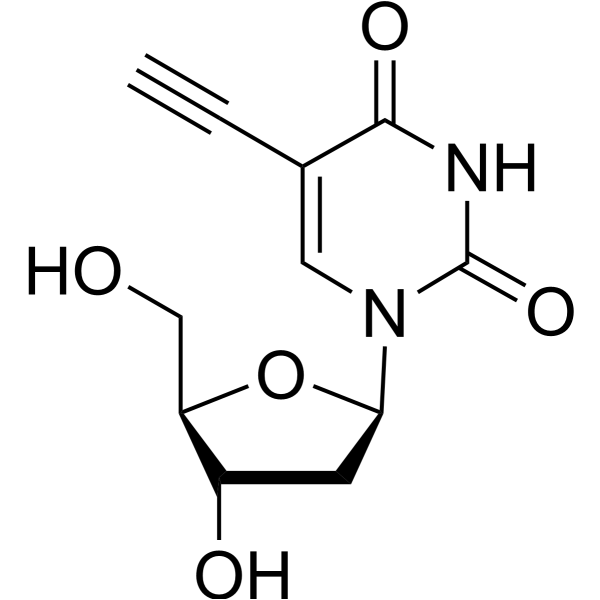
-
- HY-130801
-
|
5-OHdU
|
Endogenous Metabolite
|
Metabolic Disease
|
|
5-Hydroxy-2'-deoxyuridine (5-OHdU) is a major stable oxidation product of 2'-Deoxycytidine. 5-Hydroxy-2'-deoxyuridine can be incorporated into DNA in vitro by DNA polymerase .
|
-
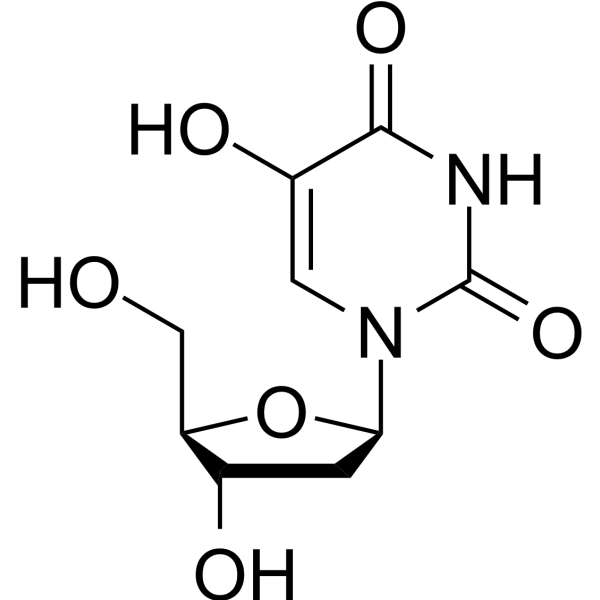
-
- HY-135957
-
|
N3dUrd
|
DNA/RNA Synthesis
|
Cancer
|
|
2'-Azido-2'-deoxyuridine (N3dUrd) is a ribonucleotide reductase inhibitor. 2'-Azido-2'-deoxyuridine has anti-cancer activity . 2'-Azido-2'-deoxyuridine is a click chemistry reagent, it contains an Azide group and can undergo copper-catalyzed azide-alkyne cycloaddition reaction (CuAAc) with molecules containing Alkyne groups. Strain-promoted alkyne-azide cycloaddition (SPAAC) can also occur with molecules containing DBCO or BCN groups.
|
-
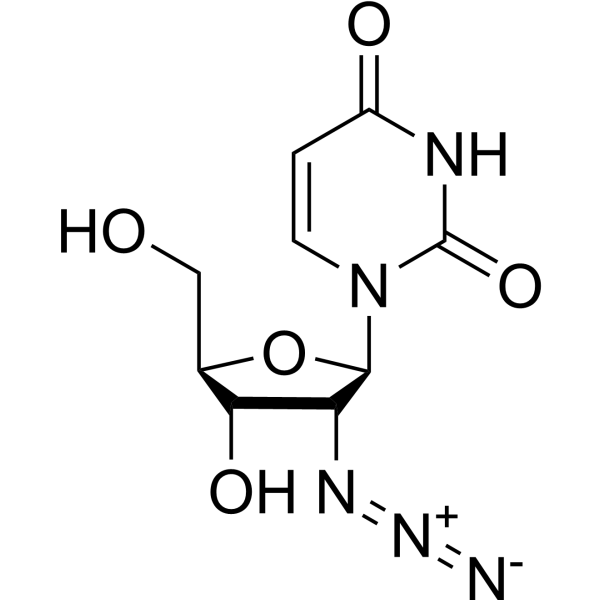
-
- HY-112669
-
|
5-ChloroDeoxyuridine; CldU
|
Others
|
Neurological Disease
|
|
5-Chloro-2'-deoxyuridine, a thymine analog, is to study the potential of hypochlorous acid damage to DNA and DNA precursors.
|
-
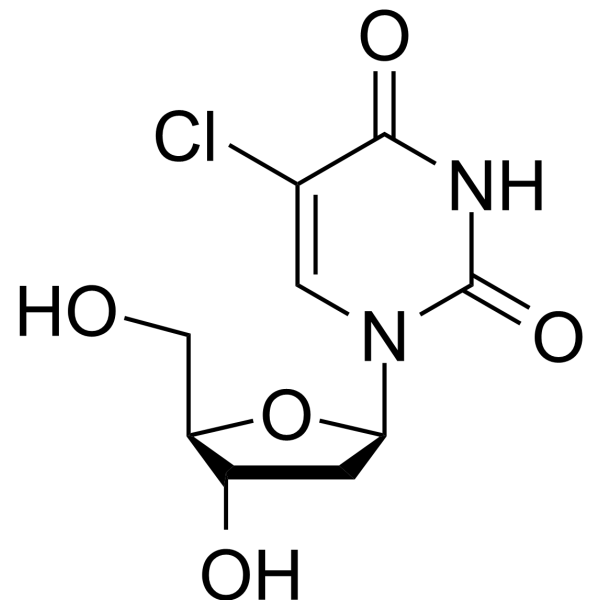
-
- HY-W154172
-
|
|
Nucleoside Antimetabolite/Analog
|
Cancer
|
|
2′-Chloro-2′-deoxyuridine is a purine nucleoside analog. Purine nucleoside analogs have broad antitumor activity targeting indolent lymphoid malignancies. Anticancer mechanisms in this process rely on inhibition of DNA synthesis, induction of apoptosis, etc .
|
-
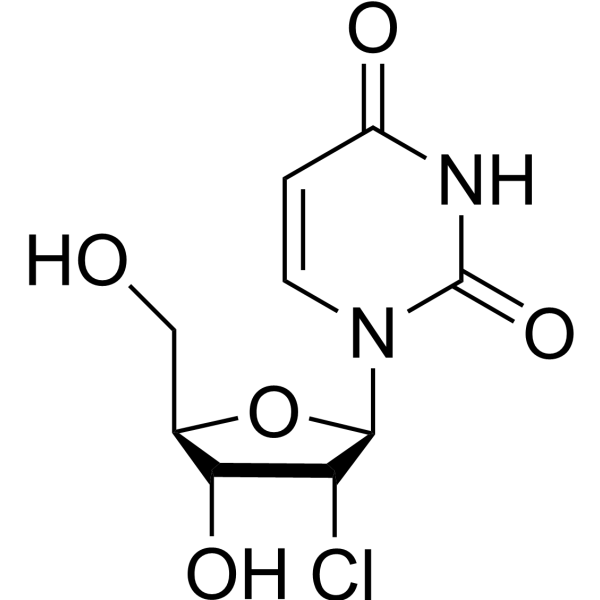
-
- HY-154120
-
|
|
Nucleoside Antimetabolite/Analog
|
Cancer
|
|
6-Aza-2'-deoxyuridine is a purine nucleoside analog. Purine nucleoside analogs have broad antitumor activity targeting indolent lymphoid malignancies. Anticancer mechanisms in this process rely on inhibition of DNA synthesis, induction of apoptosis, etc .
|
-
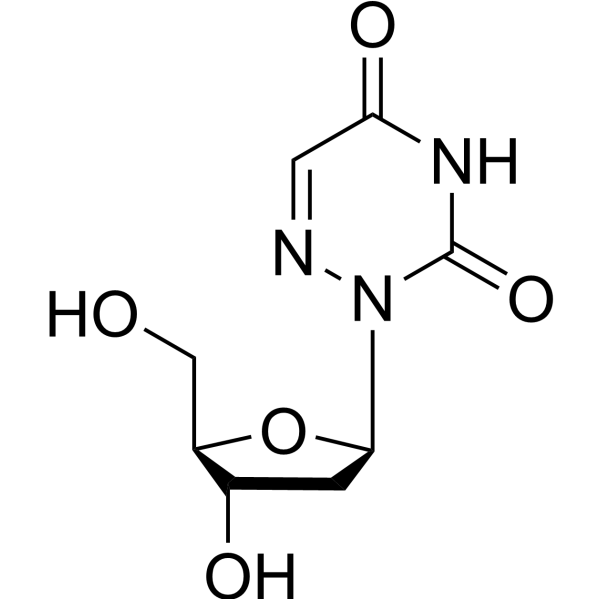
-
- HY-154533
-
|
|
Nucleoside Antimetabolite/Analog
|
Cancer
|
|
3’-O-Acetyl-2’-deoxyuridine is a purine nucleoside analog. Purine nucleoside analogs have broad antitumor activity targeting indolent lymphoid malignancies. Anticancer mechanisms in this process rely on inhibition of DNA synthesis, induction of apoptosis, etc .
|
-
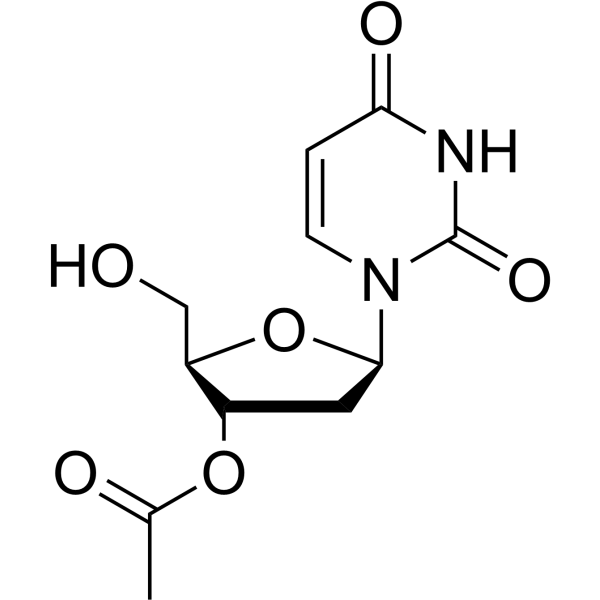
-
- HY-154239
-
|
|
Nucleoside Antimetabolite/Analog
|
Cancer
|
|
5’-O-Triphenylmethyl-2’-deoxyuridine is a purine nucleoside analog. Purine nucleoside analogs have broad antitumor activity targeting indolent lymphoid malignancies. Anticancer mechanisms in this process rely on inhibition of DNA synthesis, induction of apoptosis, etc .
|
-
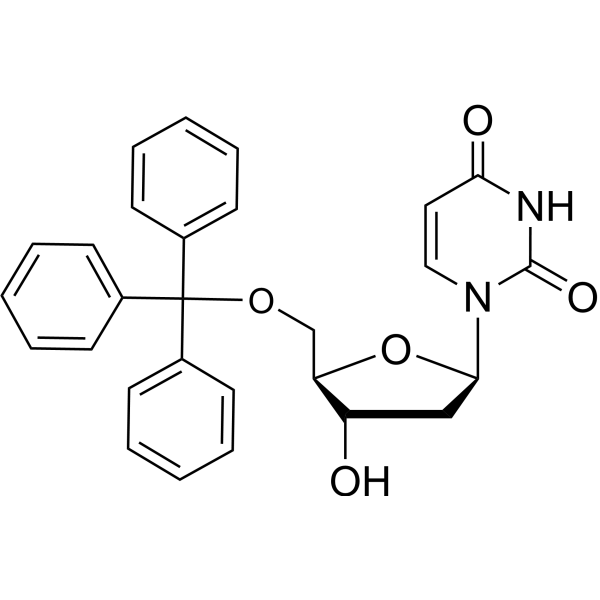
-
- HY-154026
-
|
|
Nucleoside Antimetabolite/Analog
|
Cancer
|
|
5-Amino-2'-deoxyuridine (monohydrochloride) is a purine nucleoside analog. Purine nucleoside analogs have broad antitumor activity targeting indolent lymphoid malignancies. Anticancer mechanisms in this process rely on inhibition of DNA synthesis, induction of apoptosis, etc .
|
-
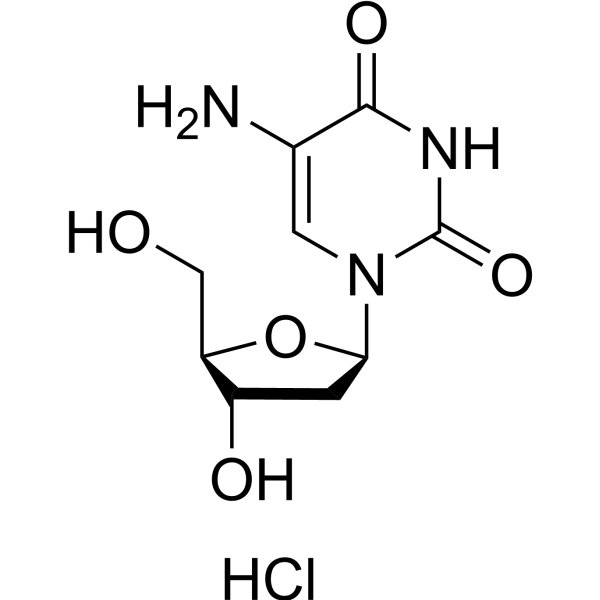
-
- HY-48873
-
|
|
DNA/RNA Synthesis
|
Others
|
|
5'-O-DMT-5-Ethynyl-2'-deoxyuridine 3'-CE phosphoramidite is a phosphorite monomer that can be used in the synthesis of oligonucleotides.
|
-
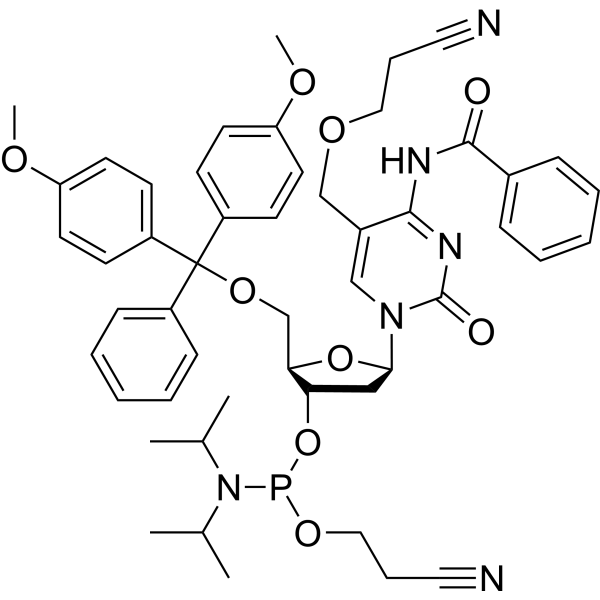
-
- HY-W097792
-
|
5'-O-DMT-dU
|
DNA/RNA Synthesis
|
Others
|
|
5'-O-(4,4'-Dimethoxytrityl)-2'-deoxyuridine (5'-O-DMT-dU) is a competitive inhibitor of E. coli dUTP nucleotidohydrolase (dUTPase), with the Ki higher than 1000 μM. 5'-O-(4,4'-Dimethoxytrityl)-2'-deoxyuridine can be used in machine-assisted DNA synthesis by synthesizing nucleosidic phosphoramidite blocks [2].
|
-
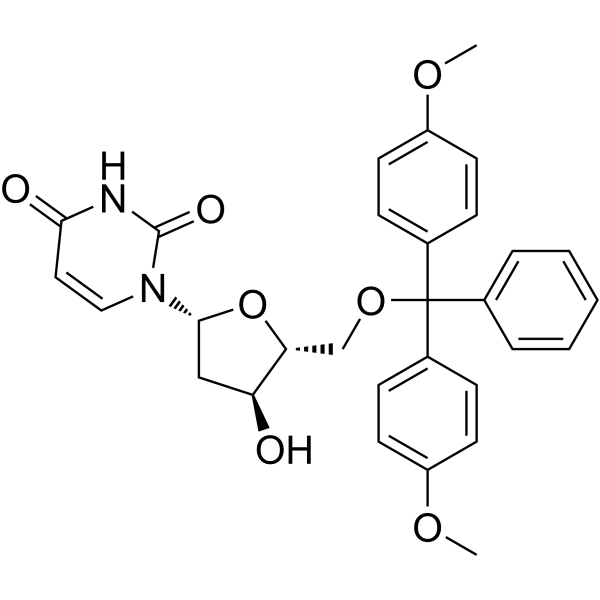
-
- HY-W553730
-
|
|
Nucleoside Antimetabolite/Analog
|
Cancer
|
|
2’-Deoxy-2’-iodouridine; 2’-Iodo-2’-deoxyuridine is a purine nucleoside analog. Purine nucleoside analogs have broad antitumor activity targeting indolent lymphoid malignancies. Anticancer mechanisms in this process rely on inhibition of DNA synthesis, induction of apoptosis, etc .
|
-
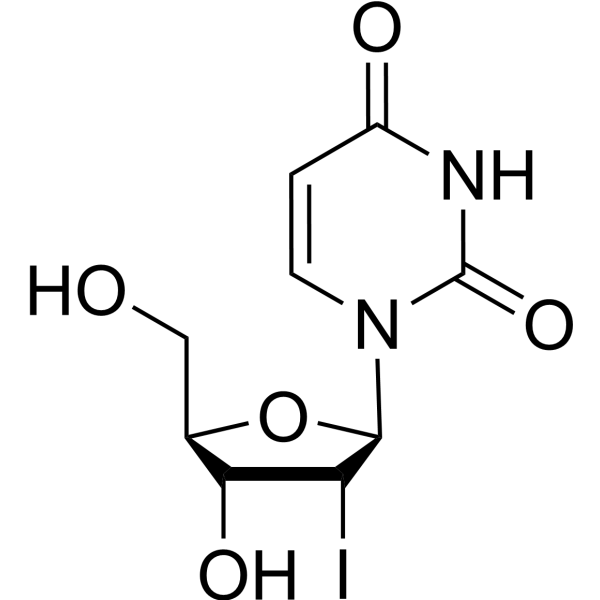
-
- HY-W141338
-
|
2'-Bromo-2'-deoxy-D-uridine
|
Nucleoside Antimetabolite/Analog
|
Cancer
|
|
2′-Bromo-2′-deoxyuridine is a purine nucleoside analog. Purine nucleoside analogs have broad antitumor activity targeting indolent lymphoid malignancies. Anticancer mechanisms in this process rely on inhibition of DNA synthesis, induction of apoptosis, etc .
|
-
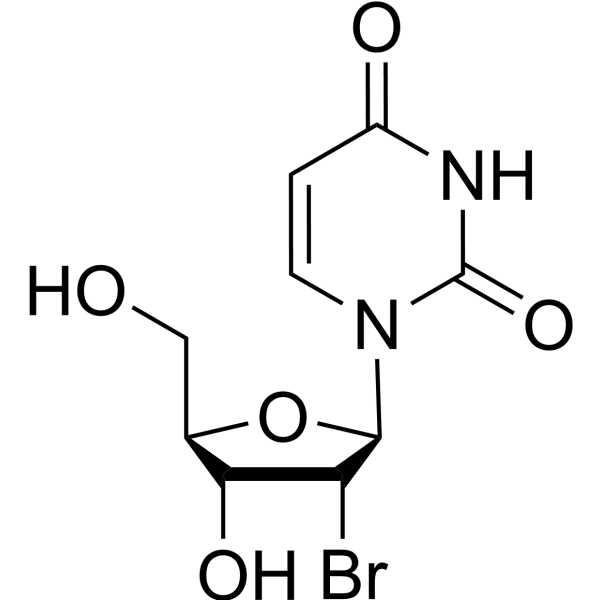
-
- HY-W251781
-
|
2′-Deoxy-4-thiouridine; 4-ThioDeoxyuridine
|
Nucleoside Antimetabolite/Analog
|
Cancer
|
|
4-Thio-2’-deoxyuridine is a purine nucleoside analog. Purine nucleoside analogs have broad antitumor activity targeting indolent lymphoid malignancies. Anticancer mechanisms in this process rely on inhibition of DNA synthesis, induction of apoptosis, etc .
|
-
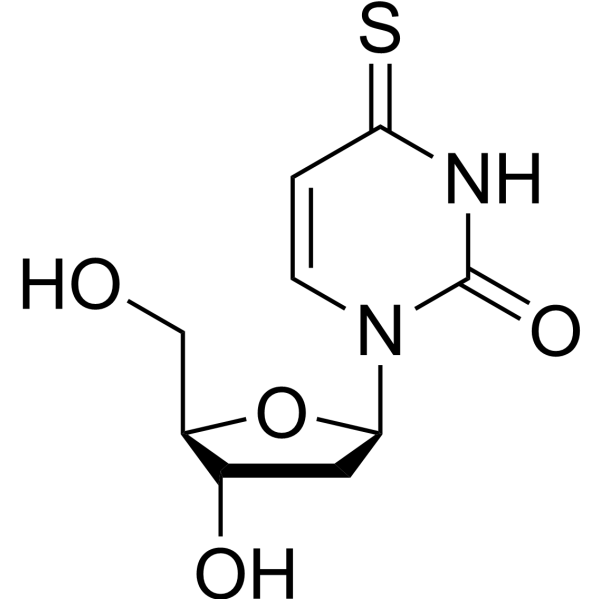
-
- HY-154423
-
|
|
Nucleoside Antimetabolite/Analog
|
Cancer
|
|
2′-Azido-5′-O-[bis(4-methoxyphenyl)phenylmethyl]-2′-deoxyuridine is a purine nucleoside analog. Purine nucleoside analogs have broad antitumor activity targeting indolent lymphoid malignancies. Anticancer mechanisms in this process rely on inhibition of DNA synthesis, induction of apoptosis, etc . 2′-Azido-5′-O-[bis(4-methoxyphenyl)phenylmethyl]-2′-deoxyuridine is a click chemistry reagent, it contains an Azide group and can undergo copper-catalyzed azide-alkyne cycloaddition reaction (CuAAc) with molecules containing Alkyne groups. Strain-promoted alkyne-azide cycloaddition (SPAAC) can also occur with molecules containing DBCO or BCN groups.
|
-
![<em>2</em>′-Azido-5′-O-[bis(4-methoxyphenyl)phenylmethyl]-<em>2′-deoxyuridine</em>](//file.medchemexpress.com/product_pic/hy-154423.gif)
-
- HY-B0307A
-
|
5-Iodo-2′-Deoxyuridine hydrate; 5-IUdR hydrate; IdUrd hydrate
|
Phosphatase
|
Infection
|
|
Idoxuridine (5-Iodo-2′-deoxyuridine, 5-IUdR, IdUrd) hydrate is an iodinated thymidine analogue that competitively inhibits phosphorylases. Idoxuridine can inhibit viral activity, particularly viral eye infections, including herpes simplex keratitis, by inhibiting DNA polymerase and affecting viral replication. Idoxuridine against feline herpesvirus has the IC50 value of 4.3 μM .
|
-
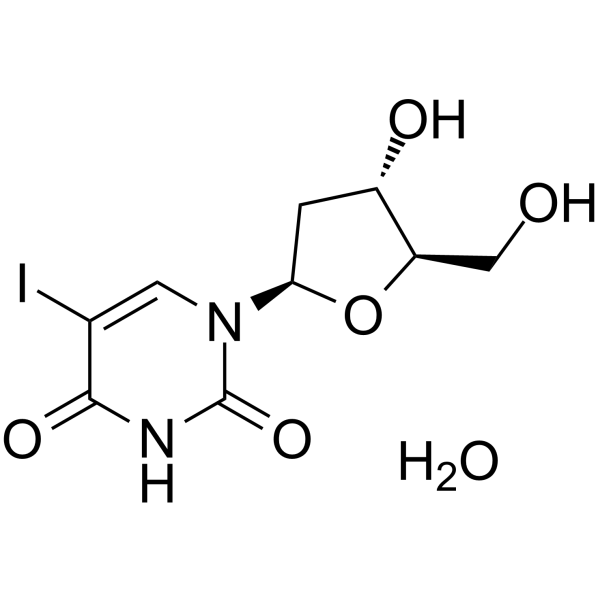
-
- HY-154160
-
|
|
Nucleoside Antimetabolite/Analog
|
Cancer
|
|
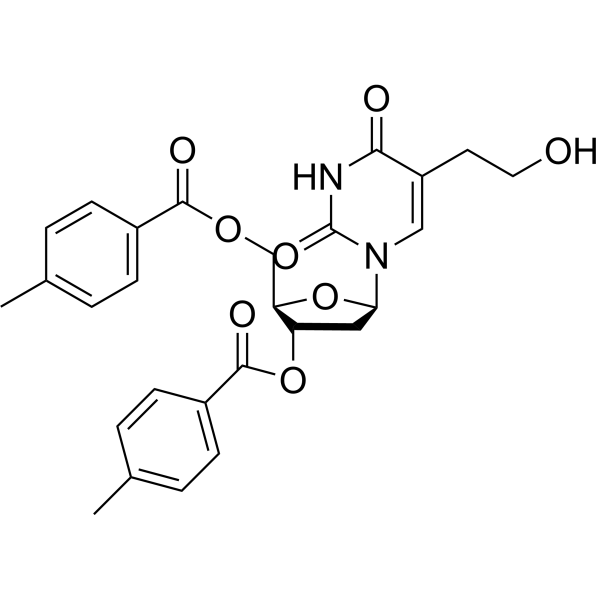
5-(2-Hydroxyethyl)-2’,3’-di-O-toluoyl-2’-deoxyuridine is a purine nucleoside analog. Purine nucleoside analogs have broad antitumor activity targeting indolent lymphoid malignancies. Anticancer mechanisms in this process rely on inhibition of DNA synthesis, induction of apoptosis, etc .
|
-
-
- HY-B0307
-
|
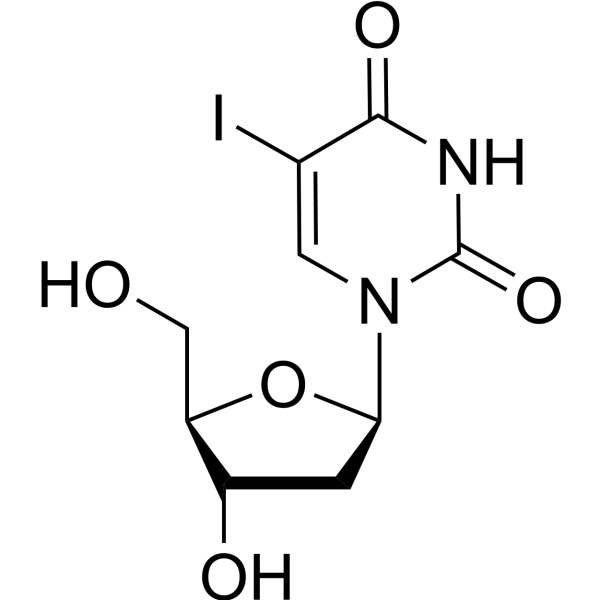
5-Iodo-2′-Deoxyuridine; 5-IUdR; IdUrd
|
Phosphatase
Orthopoxvirus
|
Infection
Cancer
|
|
Idoxuridine (5-Iodo-2′-deoxyuridine, 5-IUdR, IdUrd) is an iodinated thymidine analogue that competitively inhibits phosphorylases. Idoxuridine can inhibit viral activity, particularly viral eye infections, including herpes simplex keratitis, by inhibiting DNA polymerase and affecting viral replication. Idoxuridine against feline herpesvirus has the IC50 value of 4.3 μM . Idoxuridine shows anti-orthopoxvirus activity.
|
-
-
- HY-154424
-
|
|
Nucleoside Antimetabolite/Analog
|
Cancer
|
|
2′-Amino-5′-O-[bis(4-methoxyphenyl)phenylmethyl]-2′-deoxyuridine is a purine nucleoside analog. Purine nucleoside analogs have broad antitumor activity targeting indolent lymphoid malignancies. Anticancer mechanisms in this process rely on inhibition of DNA synthesis, induction of apoptosis, etc .
|
-
![<em>2</em>′-Amino-5′-O-[bis(4-methoxyphenyl)phenylmethyl]-<em>2′-deoxyuridine</em>](//file.medchemexpress.com/product_pic/hy-154424.gif)
-
- HY-W397503
-
|
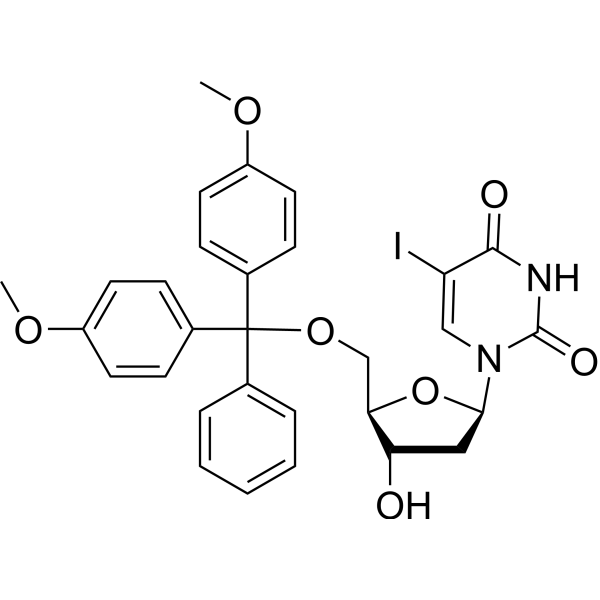
2'-Deoxy-5'-O-DMT-5-iodouridine
|
Nucleoside Antimetabolite/Analog
|
Cancer
|
|
5’-O-DMTr-5-iodo-2’-deoxyuridine is a purine nucleoside analog. Purine nucleoside analogs have broad antitumor activity targeting indolent lymphoid malignancies. Anticancer mechanisms in this process rely on inhibition of DNA synthesis, induction of apoptosis, etc .
|
-
-
- HY-138253S
-
|
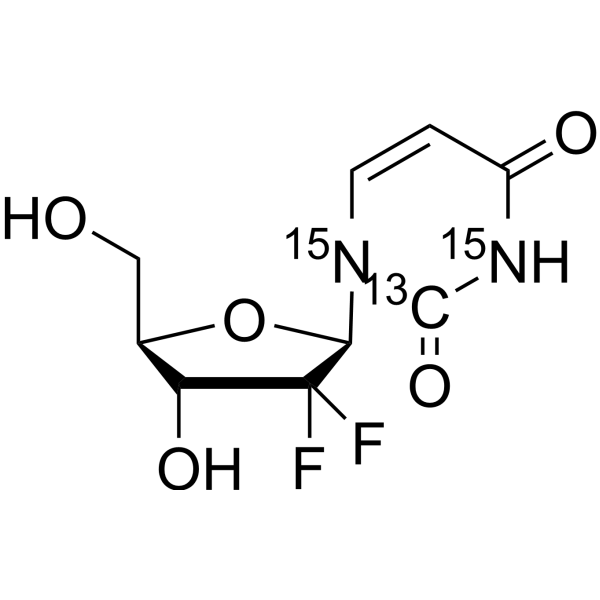
dFdU-13C,15N2; 2',2'-Difluoro-2'-Deoxyuridine-13C,15N2
|
Isotope-Labeled Compounds
|
Others
|
|
2′,2′-Difluorodeoxyuridine- 13C, 15N2 (dFdU- 13C, 15N2; 2',2'-Difluoro-2'-deoxyuridine- 13C, 15N2) is a 13C- and 15N-labeled compound.
|
-
-
- HY-W017068A
-
-
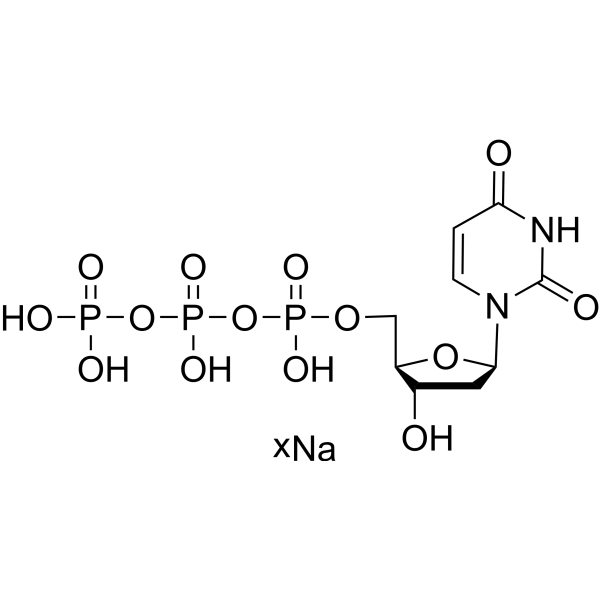
-
- HY-154211
-
|
|
DNA/RNA Synthesis
|
Others
|
|
5’-O-DMTr-dU-methyl phosphonamidite; 5’-O-DMTr-2’-deoxyuridine-3’-O-(P-methyl-N,N-diisopropylamino)phosphonamidite is a phosphoramidite that can be used in the synthesis of oligonucleotides.
|
-
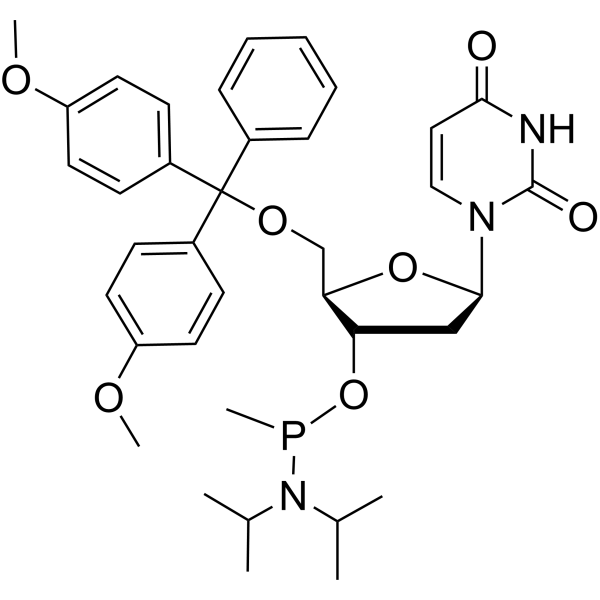
-
- HY-W017068
-
-
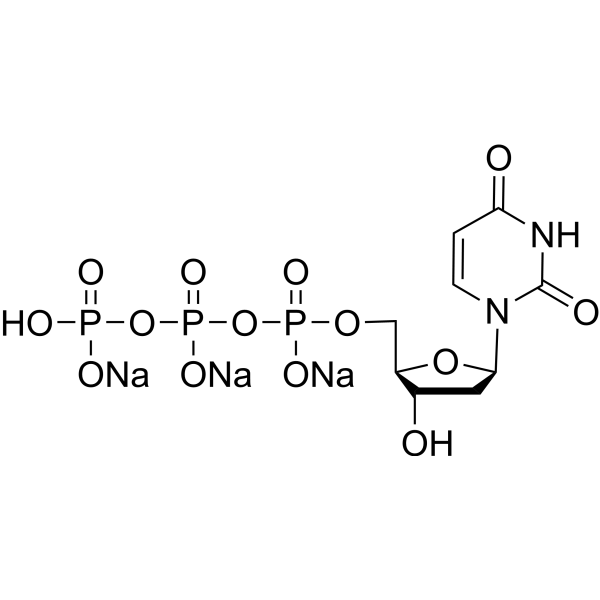
-
- HY-15910
-
5-BrdU
Maximum Cited Publications
47 Publications Verification
BrdU; 5-Bromo-2'-Deoxyuridine; BUdR
|
Nucleoside Antimetabolite/Analog
DNA/RNA Synthesis
|
Cancer
|
|
5-BrdU (BrdU) is a nucleoside analog that competes with thymidine for incorporation into DNA. 5-BrdU is commonly used in the detection of proliferating cells.
|
-
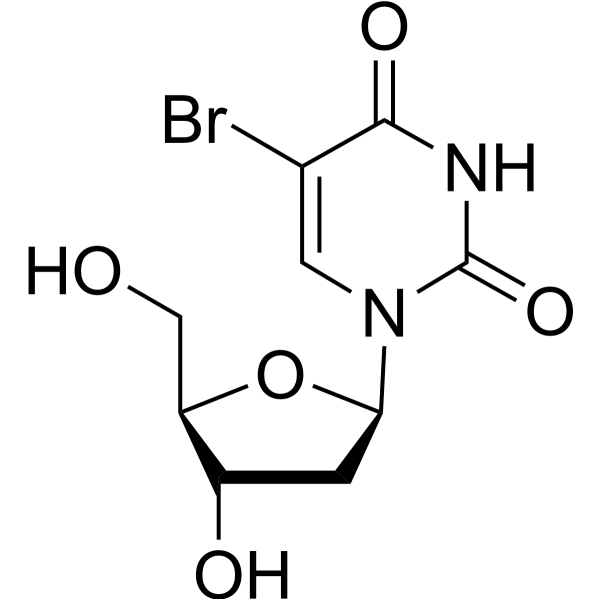
-
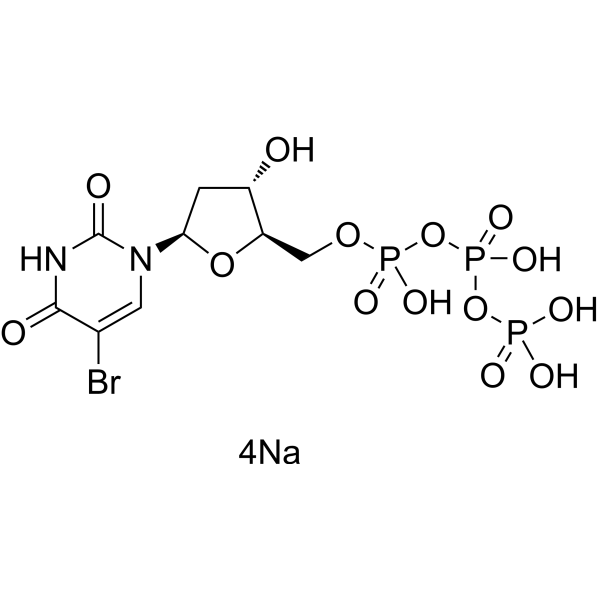
- HY-D1023
-
|
5-Bromo-2'-Deoxyuridine 5'-triphosphate sodium salt
|
DNA/RNA Synthesis
|
Others
|
|
5-BrdUTP sodium salt is a TdT substrate which can be used to label the DNA double-strand breaks.
|
-
-
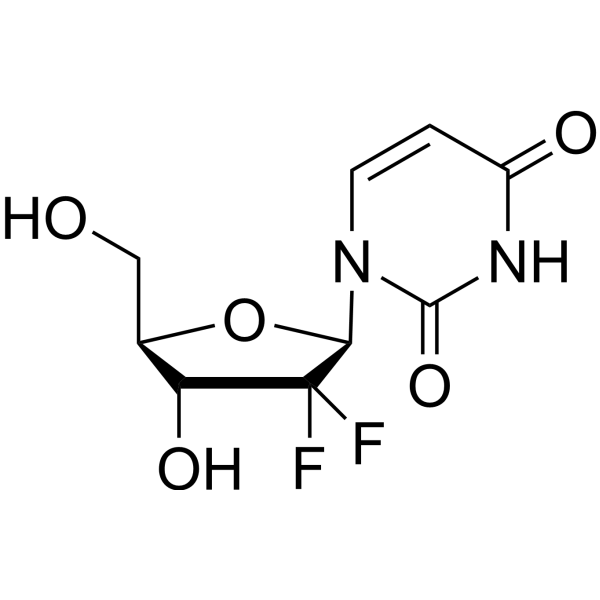
- HY-138253
-
|
dFdU; 2',2'-Difluoro-2'-Deoxyuridine
|
Drug Metabolite
|
Cancer
|
|
2′,2′-Difluorodeoxyuridine (dFdU) is the main metabolite of Gemcitabine (HY-17026). 2′,2′-Difluorodeoxyuridine causes a concentration- and schedule- dependent radiosensitising effect in vitro .
|
-
-
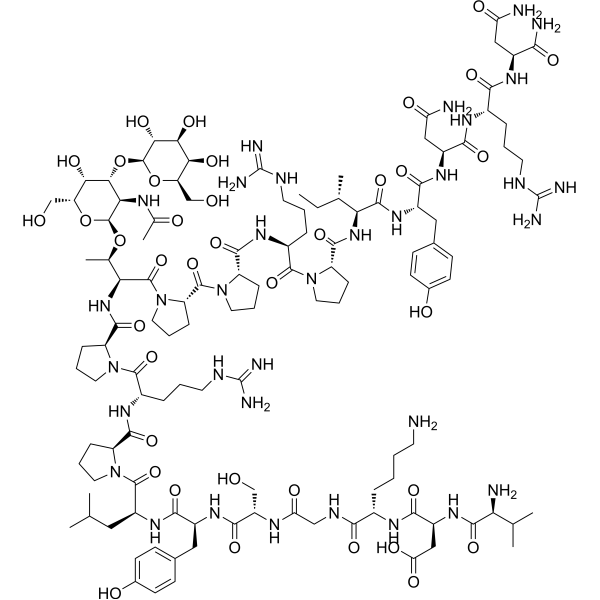
- HY-P5443
-
|
|
Bacterial
|
Others
|
|
Pyrrhocoricin is a biological active peptide. (Antimicrobial activity against Gram-negative bacteria)
|
-
| Cat. No. |
Product Name |
Type |
-
- HY-W097792
-
|
5'-O-DMT-dU
|
Gene Sequencing and Synthesis
|
|
5'-O-(4,4'-Dimethoxytrityl)-2'-deoxyuridine (5'-O-DMT-dU) is a competitive inhibitor of E. coli dUTP nucleotidohydrolase (dUTPase), with the Ki higher than 1000 μM. 5'-O-(4,4'-Dimethoxytrityl)-2'-deoxyuridine can be used in machine-assisted DNA synthesis by synthesizing nucleosidic phosphoramidite blocks [2].
|
-
- HY-D1023
-
|
5-Bromo-2'-Deoxyuridine 5'-triphosphate sodium salt
|
Biochemical Assay Reagents
|
|
5-BrdUTP sodium salt is a TdT substrate which can be used to label the DNA double-strand breaks.
|
| Cat. No. |
Product Name |
Target |
Research Area |
-
- HY-P5443
-
|
|
Bacterial
|
Others
|
|
Pyrrhocoricin is a biological active peptide. (Antimicrobial activity against Gram-negative bacteria)
|
| Cat. No. |
Product Name |
Category |
Target |
Chemical Structure |
| Cat. No. |
Product Name |
Chemical Structure |
-
- HY-D0186S5
-
|
|
|
2'-Deoxyuridine-d is the deuterium labeled 2'-Deoxyuridine. 2'-Deoxyuridine could increase chromosome breakage and results in a decreased thymidylate synthetase activity. A known use of 2'-Deoxyuridine is as a precursor in the synthesis of Edoxud[1]<
|
-

-
- HY-D0186S4
-
|
|
|
2'-Deoxyuridine-d2 is the deuterium labeled 2'-Deoxyuridine. 2'-Deoxyuridine could increase chromosome breakage and results in a decreased thymidylate synthetase activity. A known use of 2'-Deoxyuridine is as a precursor in the synthesis of Edoxud[1]
|
-

-
- HY-D0186S1
-
|
|
|
2'-Deoxyuridine-2′- 13C is the 13C labeled 2'-Deoxyuridine. 2'-Deoxyuridine could increase chromosome breakage and results in a decreased thymidylate synthetase activity. A known use of 2'-Deoxyuridine is as a precursor in the synthesis of Edoxud[1]
|
-

-
- HY-D0186S
-
|
|
|
2'-Deoxyuridine-1′- 13C is the 13C labeled 2'-Deoxyuridine. 2'-Deoxyuridine could increase chromosome breakage and results in a decreased thymidylate synthetase activity. A known use of 2'-Deoxyuridine is as a precursor in the synthesis of Edoxud[1]
|
-

-
- HY-D0186S2
-
|
|
|
2'-Deoxyuridine-3′- 13C is the 13C labeled 2'-Deoxyuridine. 2'-Deoxyuridine could increase chromosome breakage and results in a decreased thymidylate synthetase activity. A known use of 2'-Deoxyuridine is as a precursor in the synthesis of Edoxud[1]
|
-

-
- HY-D0186S3
-
|
|
|
2'-Deoxyuridine-5′- 13C is the 13C labeled 2'-Deoxyuridine. 2'-Deoxyuridine could increase chromosome breakage and results in a decreased thymidylate synthetase activity. A known use of 2'-Deoxyuridine is as a precursor in the synthesis of Edoxud[1]
|
-

-
- HY-D0186S8
-
|
|
|
2'-Deoxyuridine-d2-1 is the deuterium labeled 2'-Deoxyuridine[1]. 2'-Deoxyuridine could increase chromosome breakage and results in a decreased thymidylate synthetase activity. A known use of 2'-Deoxyuridine is as a precursor in the synthesis of Edoxudine[2].
|
-

-
- HY-D0186S7
-
|
|
|
2'-Deoxyuridine- 13C, 15N2 is the 13C and 15N labeled 2'-Deoxyuridine[1]. 2'-Deoxyuridine could increase chromosome breakage and results in a decreased thymidylate synthetase activity. A known use of 2'-Deoxyuridine is as a precursor in the synthesis of Edoxudine[2].
|
-

-
- HY-138253S
-
|
|
|
2′,2′-Difluorodeoxyuridine- 13C, 15N2 (dFdU- 13C, 15N2; 2',2'-Difluoro-2'-deoxyuridine- 13C, 15N2) is a 13C- and 15N-labeled compound.
|
-

| Cat. No. |
Product Name |
|
Classification |
-
- HY-118411
-
|
|
|
Alkynes
PROTAC Synthesis
|
|
5-Ethynyl-2'-deoxyuridine (EdU), a thymidine analogue, is incorporated into cellular DNA during DNA replication and the subsequent reaction of EdU with a fluorescent azide in a “Click” reaction. EdU staining is a fast, sensitive and reproducible method to study cell proliferation . 5-Ethynyl-2'-deoxyuridine is an alkyl chain-based PROTAC linker that can be used in the synthesis of PROTACs [2]. 5-Ethynyl-2'-deoxyuridine is a click chemistry reagent, it contains an Alkyne group and can undergo copper-catalyzed azide-alkyne cycloaddition (CuAAc) with molecules containing Azide groups.
|
-
- HY-135957
-
|
N3dUrd
|
|
Azide
|
|
2'-Azido-2'-deoxyuridine (N3dUrd) is a ribonucleotide reductase inhibitor. 2'-Azido-2'-deoxyuridine has anti-cancer activity . 2'-Azido-2'-deoxyuridine is a click chemistry reagent, it contains an Azide group and can undergo copper-catalyzed azide-alkyne cycloaddition reaction (CuAAc) with molecules containing Alkyne groups. Strain-promoted alkyne-azide cycloaddition (SPAAC) can also occur with molecules containing DBCO or BCN groups.
|
-
- HY-154423
-
|
|
|
Azide
|
|
2′-Azido-5′-O-[bis(4-methoxyphenyl)phenylmethyl]-2′-deoxyuridine is a purine nucleoside analog. Purine nucleoside analogs have broad antitumor activity targeting indolent lymphoid malignancies. Anticancer mechanisms in this process rely on inhibition of DNA synthesis, induction of apoptosis, etc . 2′-Azido-5′-O-[bis(4-methoxyphenyl)phenylmethyl]-2′-deoxyuridine is a click chemistry reagent, it contains an Azide group and can undergo copper-catalyzed azide-alkyne cycloaddition reaction (CuAAc) with molecules containing Alkyne groups. Strain-promoted alkyne-azide cycloaddition (SPAAC) can also occur with molecules containing DBCO or BCN groups.
|
Your information is safe with us. * Required Fields.
Inquiry Information
- Product Name:
- Cat. No.:
- Quantity:
- MCE Japan Authorized Agent:

































![<em>2</em>′-Azido-5′-O-[bis(4-methoxyphenyl)phenylmethyl]-<em>2′-deoxyuridine</em>](http://file.medchemexpress.com/product_pic/hy-154423.gif)



![<em>2</em>′-Amino-5′-O-[bis(4-methoxyphenyl)phenylmethyl]-<em>2′-deoxyuridine</em>](http://file.medchemexpress.com/product_pic/hy-154424.gif)











