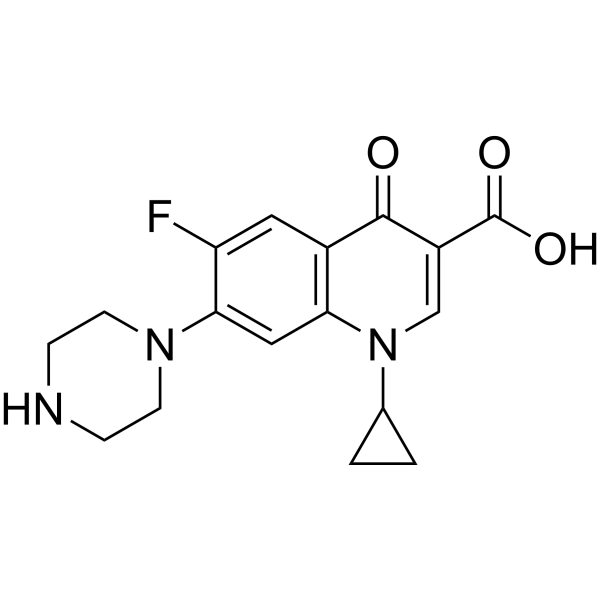
|
A-431
|
IC50 |
|
Inhibition of human A431 cell proliferation by MTT assay
Inhibition of human A431 cell proliferation by MTT assay
|
[PMID: 19595598]
|
|
A-431
|
IC50 |
137 μM
Compound: Ciprofloxacin
|
Cytotoxicity against human A431 cells assessed as reduction in cell viability by MTT assay
Cytotoxicity against human A431 cells assessed as reduction in cell viability by MTT assay
|
[PMID: 30660827]
|
|
A-431
|
IC50 |
|
Anticancer activity against human A-431 cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
Anticancer activity against human A-431 cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
|
[PMID: 31881454]
|
|
A549
|
IC50 |
|
Antitumor activity against human A549 cells assessed as reduction in cell viability incubated for 4 hrs by MTT assay
Antitumor activity against human A549 cells assessed as reduction in cell viability incubated for 4 hrs by MTT assay
|
[PMID: 31881454]
|
|
A549
|
IC50 |
|
Antitumor activity against human A549 cells after 5 days by MTT assay
Antitumor activity against human A549 cells after 5 days by MTT assay
|
[PMID: 19595598]
|
|
A549
|
IC50 |
> 100 μM
Compound: Ciprofloxacin
|
Antiproliferative activity against human A549 cells after 24 hrs by BrdU incorporation assay
Antiproliferative activity against human A549 cells after 24 hrs by BrdU incorporation assay
|
[PMID: 27555286]
|
|
A549
|
IC50 |
> 302 nM
Compound: Ciprofloxacin
|
Antiproliferative activity against human A549 cells after 72 hrs by MTT assay
Antiproliferative activity against human A549 cells after 72 hrs by MTT assay
|
[PMID: 30660827]
|
|
B-cell
|
IC50 |
123 μM
Compound: ciprofloxacin
|
Antiproliferative activity against Theileria parva-induced proliferation of bovine B lymphocyte assessed as inhibition of [3H]thymidine uptake after 48 hrs
Antiproliferative activity against Theileria parva-induced proliferation of bovine B lymphocyte assessed as inhibition of [3H]thymidine uptake after 48 hrs
|
[PMID: 19075064]
|
|
C8166
|
EC50 |
100 μM
Compound: CPX, ciprofloxacin
|
In vitro antiviral activity against HIV-1 IIIB in C8166 (human CD4) cells.
In vitro antiviral activity against HIV-1 IIIB in C8166 (human CD4) cells.
|
[PMID: 11020296]
|
|
CAPAN-1
|
IC50 |
> 100 μM
Compound: Ciprofloxacin
|
Cytotoxicity against human Capan1 cells assessed as reduction in cell viability by MTT assay
Cytotoxicity against human Capan1 cells assessed as reduction in cell viability by MTT assay
|
[PMID: 30660827]
|
|
CHO
|
CC50 |
|
Cytotoxicity against CHO cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
Cytotoxicity against CHO cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
|
[PMID: 31525660]
|
|
CHO
|
IC50 |
|
Cytotoxicity against Chinese hamster CHO cells assessed as reduction in cell viability by MTT assay
Cytotoxicity against Chinese hamster CHO cells assessed as reduction in cell viability by MTT assay
|
[PMID: 31881454]
|
|
CHO
|
IC50 |
> 150 μM
Compound: Ciprofloxacin
|
Cytotoxicity against CHO cells assessed as reduction in cell viability
Cytotoxicity against CHO cells assessed as reduction in cell viability
|
[PMID: 30660827]
|
|
CT26
|
IC50 |
|
Antitumor activity against mouse CT26 cells assessed as reduction in cell viability incubated for 4 hrs by MTT assay
Antitumor activity against mouse CT26 cells assessed as reduction in cell viability incubated for 4 hrs by MTT assay
|
[PMID: 31881454]
|
|
EJ
|
IC50 |
137 μM
Compound: Ciprofloxacin
|
Cytotoxicity against human EJ cells assessed as reduction in cell viability by MTT assay
Cytotoxicity against human EJ cells assessed as reduction in cell viability by MTT assay
|
[PMID: 30660827]
|
|
EJ
|
IC50 |
|
Inhibition of human EJ cell proliferation by MTT assay
Inhibition of human EJ cell proliferation by MTT assay
|
[PMID: 19595598]
|
|
EJ
|
IC50 |
|
Anticancer activity against human EJ cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
Anticancer activity against human EJ cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
|
[PMID: 31881454]
|
|
HBL-100
|
IC50 |
10.28 μM
Compound: C; Cip
|
Cytotoxicity against human HBL-100 cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
Cytotoxicity against human HBL-100 cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
|
[PMID: 34319100]
|
|
HEK-293T
|
CC50 |
> 60 μg/mL
Compound: Ciprofloxacin
|
Cytotoxicity against HEK293T cells assessed as reduction in cell viability measured after 24 hrs by MTT assay
Cytotoxicity against HEK293T cells assessed as reduction in cell viability measured after 24 hrs by MTT assay
|
[PMID: 27720324]
|
|
HEp-2
|
CC50 |
|
Cytotoxicity against human Hep2 cell line after 72 hrs
Cytotoxicity against human Hep2 cell line after 72 hrs
|
[PMID: 17228862]
|
|
HEp-2
|
CC50 |
|
Cytotoxicity against human Hep2 cells after 72 hrs by alamar blue assay
Cytotoxicity against human Hep2 cells after 72 hrs by alamar blue assay
|
[PMID: 21425851]
|
|
HEp-2
|
CC50 |
|
Cytotoxicity against human Hep2 cell line after 72 hrs
Cytotoxicity against human Hep2 cell line after 72 hrs
|
[PMID: 21443219]
|
|
HEp-2
|
CC50 |
> 100 μM
Compound: Ciprofloxacin
|
Cytotoxicity against Hep2 cell line
Cytotoxicity against Hep2 cell line
|
[PMID: 16337789]
|
|
HEp-2
|
CC50 |
> 100 μM
Compound: Ciprofloxacin
|
Cytotoxicity against human Hep2 laryngeal carcinoma cell line
Cytotoxicity against human Hep2 laryngeal carcinoma cell line
|
[PMID: 16337791]
|
|
HL-60
|
IC50 |
> 100 μM
Compound: Ciprofloxacin
|
Cytotoxicity against human HL60 cells assessed as reduction in cell viability by MTT assay
Cytotoxicity against human HL60 cells assessed as reduction in cell viability by MTT assay
|
[PMID: 30660827]
|
|
HL-60
|
IC50 |
|
Cytotoxicity against human HL-60 cells assessed as reduction in cell viability by MTT assay
Cytotoxicity against human HL-60 cells assessed as reduction in cell viability by MTT assay
|
[PMID: 31881454]
|
|
HL-60
|
IC50 |
> 150 μM
Compound: Ciprofloxacin
|
Cytotoxicity against human HL60 cells assessed as reduction in cell viability
Cytotoxicity against human HL60 cells assessed as reduction in cell viability
|
[PMID: 30660827]
|
|
HL-60
|
IC50 |
> 302 nM
Compound: Ciprofloxacin
|
Antiproliferative activity against human HL60 cells after 72 hrs by MTT assay
Antiproliferative activity against human HL60 cells after 72 hrs by MTT assay
|
[PMID: 30660827]
|
|
HL-60
|
IC50 |
>= 100 μM
Compound: Ciprofloxacin
|
Antiproliferative activity against human HL60 cells by MTT assay
Antiproliferative activity against human HL60 cells by MTT assay
|
[PMID: 30660827]
|
|
HaCaT
|
IC50 |
|
Cytotoxicity against human HaCaT cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
Cytotoxicity against human HaCaT cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
|
[PMID: 31678743]
|
|
HeLa
|
EC50 |
0.03 μM
Compound: Ciprofloxacin
|
Inhibitory activity against HeLa cell Topoisomerase II
Inhibitory activity against HeLa cell Topoisomerase II
|
10.1016/0960-894X(95)00160-U
|
|
HeLa
|
IC50 |
120 μg/mL
Compound: Ciprofloxacin
|
Inhibition of metabolic activity in HeLa cells assessed as MTT reduction after 48 hrs
Inhibition of metabolic activity in HeLa cells assessed as MTT reduction after 48 hrs
|
[PMID: 17088489]
|
|
HeLa
|
IC50 |
|
Inhibition of human HeLa cell proliferation
Inhibition of human HeLa cell proliferation
|
[PMID: 19595598]
|
|
HeLa
|
IC50 |
290 μg/mL
Compound: Ciprofloxacin
|
Antiproliferative effect against HeLa cells after 48 hrs
Antiproliferative effect against HeLa cells after 48 hrs
|
[PMID: 17088489]
|
|
HeLa
|
IC50 |
|
Inhibition of human HeLa cell proliferation assessed as bromodeoxyuridine incorporation during DNA synthesis after 48 hrs by ELISA
Inhibition of human HeLa cell proliferation assessed as bromodeoxyuridine incorporation during DNA synthesis after 48 hrs by ELISA
|
[PMID: 19595598]
|
|
HeLa
|
IC50 |
> 100 μM
Compound: Ciprofloxacin
|
Cytotoxicity against human HeLa cells after 24 hrs by LDH release assay
Cytotoxicity against human HeLa cells after 24 hrs by LDH release assay
|
[PMID: 27018907]
|
|
Hep 3B2
|
IC50 |
> 100 μM
Compound: Ciprofloxacin
|
Cytotoxicity against human Hep3B cells assessed as reduction in cell viability by MTT assay
Cytotoxicity against human Hep3B cells assessed as reduction in cell viability by MTT assay
|
[PMID: 30660827]
|
|
HepG2
|
CC50 |
138.6 μg/mL
Compound: CPX
|
Cytotoxicity in human HepG2 cells assessed as reduction in cell viability incubated for 24 hrs by ATP bioluminescence assay
Cytotoxicity in human HepG2 cells assessed as reduction in cell viability incubated for 24 hrs by ATP bioluminescence assay
|
[PMID: 30067360]
|
|
HepG2
|
CC50 |
|
Cytotoxicity against human HepG2 cells after 72 hrs
Cytotoxicity against human HepG2 cells after 72 hrs
|
[PMID: 17228862]
|
|
HepG2
|
IC50 |
27 μM
Compound: Ciprofloxacin
|
Cytotoxicity against human HepG2 cells assessed as reduction in cell viability after 7 days by MTT assay
Cytotoxicity against human HepG2 cells assessed as reduction in cell viability after 7 days by MTT assay
|
[PMID: 28237557]
|
|
HepG2
|
IC50 |
37.47 μg/mL
Compound: Ciprofloxacin (CPFX)
|
Cytotoxicity was determined for the compound in HepG2 cell line
Cytotoxicity was determined for the compound in HepG2 cell line
|
[PMID: 14980680]
|
|
HepG2
|
IC50 |
> 0.5 mM
Compound: CPX, B-H
|
Cytotoxicity against human HepG2 cells measured after overnight incubation by MTT assay
Cytotoxicity against human HepG2 cells measured after overnight incubation by MTT assay
|
[PMID: 21855181]
|
|
HepG2
|
IC50 |
> 128 μg/mL
Compound: CIP
|
Cytotoxicity against human HepG2 cells after 24 hrs by cell titre-glo assay
Cytotoxicity against human HepG2 cells after 24 hrs by cell titre-glo assay
|
[PMID: 27499455]
|
|
HepG2
|
IC50 |
> 128 μg/mL
Compound: CIP
|
Cytotoxicity against human HepG2 cells assessed as reduction in cell viability for 24 hrs by Celltiter-Glo assay
Cytotoxicity against human HepG2 cells assessed as reduction in cell viability for 24 hrs by Celltiter-Glo assay
|
[PMID: 35231579]
|
|
HepG2
|
IC50 |
> 170 μg/mL
Compound: Ciprofloxacin
|
Cytotoxicity against human HepG2 cells incubated for 40 hrs by celltiter-glo luminescent cell viability assay
Cytotoxicity against human HepG2 cells incubated for 40 hrs by celltiter-glo luminescent cell viability assay
|
[PMID: 38157595]
|
|
K562
|
IC50 |
|
Inhibition of human K562 cell proliferation by sulphorodhamine B assay
Inhibition of human K562 cell proliferation by sulphorodhamine B assay
|
[PMID: 19595598]
|
|
KB
|
IC50 |
|
Inhibition of human KB cell proliferation by MTT assay
Inhibition of human KB cell proliferation by MTT assay
|
[PMID: 19595598]
|
|
KB
|
IC50 |
|
Anticancer activity against human KB cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
Anticancer activity against human KB cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
|
[PMID: 31881454]
|
|
L02
|
IC50 |
12.23 μM
Compound: Ciprofloxacin
|
Cytotoxicity against human L02 cells after 72 hrs by CCK-8 assay
Cytotoxicity against human L02 cells after 72 hrs by CCK-8 assay
|
[PMID: 35398730]
|
|
L02
|
IC50 |
4.05 μg/mL
Compound: Ciprofloxacin
|
Cytotoxicity against human L02 cells after 72 hrs by CCK-8 assay
Cytotoxicity against human L02 cells after 72 hrs by CCK-8 assay
|
[PMID: 35398730]
|
|
L02
|
IC50 |
|
Cytotoxicity against human L02 cells assessed as inhibition of cell growth incubated for 24 hrs by MTT assay
Cytotoxicity against human L02 cells assessed as inhibition of cell growth incubated for 24 hrs by MTT assay
|
[PMID: 38096652]
|
|
L1210
|
IC50 |
> 100 μM
Compound: Ciprofloxacin
|
Cytotoxicity against mouse L1210 cells assessed as reduction in cell viability by MTT assay
Cytotoxicity against mouse L1210 cells assessed as reduction in cell viability by MTT assay
|
[PMID: 30660827]
|
|
L1210
|
IC50 |
|
Antiproliferative activity against mouse L1210 cells assessed as reduction in cell viability by MTT assay
Antiproliferative activity against mouse L1210 cells assessed as reduction in cell viability by MTT assay
|
[PMID: 31881454]
|
|
L1210
|
IC50 |
|
Cytotoxicity against mouse L1210 cells assessed as reduction in cell viability by MTT assay
Cytotoxicity against mouse L1210 cells assessed as reduction in cell viability by MTT assay
|
[PMID: 31881454]
|
|
L1210
|
IC50 |
> 150 μM
Compound: Ciprofloxacin
|
Cytotoxicity against mouse L1210 cells assessed as reduction in cell viability
Cytotoxicity against mouse L1210 cells assessed as reduction in cell viability
|
[PMID: 30660827]
|
|
L1210
|
IC50 |
>= 100 μM
Compound: Ciprofloxacin
|
Antiproliferative activity against mouse L1210 cells by MTT assay
Antiproliferative activity against mouse L1210 cells by MTT assay
|
[PMID: 30660827]
|
|
LoVo
|
IC50 |
|
Antitumor activity against human LoVo cells after 5 days by MTT assay
Antitumor activity against human LoVo cells after 5 days by MTT assay
|
[PMID: 19595598]
|
|
MC3T3-E1
|
IC50 |
|
Inhibition of mouse MC3T3-E1 cell proliferation after 48 to 72 hrs
Inhibition of mouse MC3T3-E1 cell proliferation after 48 to 72 hrs
|
[PMID: 19595598]
|
|
MCF-10A
|
IC50 |
> 300 μM
Compound: Ciprofloxacin
|
Cytotoxicity against human MCF10A cells after 48 hrs by WST1 assay
Cytotoxicity against human MCF10A cells after 48 hrs by WST1 assay
|
[PMID: 24513049]
|
|
MCF7
|
CC50 |
60 μg/mL
Compound: Ciprofloxacin
|
Cytotoxicity against human MCF7 cells assessed as reduction in cell viability measured after 24 hrs by MTT assay
Cytotoxicity against human MCF7 cells assessed as reduction in cell viability measured after 24 hrs by MTT assay
|
[PMID: 27720324]
|
|
MCF7
|
IC50 |
|
Inhibition of human MCF7 cell proliferation by MTT assay
Inhibition of human MCF7 cell proliferation by MTT assay
|
[PMID: 19595598]
|
|
MCF7
|
IC50 |
|
Antitumor activity against human MCF7 cells after 5 days by MTT assay
Antitumor activity against human MCF7 cells after 5 days by MTT assay
|
[PMID: 19595598]
|
|
MCF7
|
IC50 |
|
Inhibition of human MCF7 cell proliferation after 5 days by MTT assay
Inhibition of human MCF7 cell proliferation after 5 days by MTT assay
|
[PMID: 19595598]
|
|
MCF7
|
IC50 |
|
Antitumor activity against human MCF7 cells assessed as cell growth inhibition incubated for 72 hrs by MTT assay
Antitumor activity against human MCF7 cells assessed as cell growth inhibition incubated for 72 hrs by MTT assay
|
[PMID: 34319100]
|
|
MCF7
|
IC50 |
> 100 μM
Compound: Ciprofloxacin
|
Cytotoxicity against human MCF7 cells assessed as reduction in cell viability by MTT assay
Cytotoxicity against human MCF7 cells assessed as reduction in cell viability by MTT assay
|
[PMID: 30660827]
|
|
MCF7
|
IC50 |
|
Anticancer activity against human MCF7 cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
Anticancer activity against human MCF7 cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
|
[PMID: 31881454]
|
|
MG-63
|
IC50 |
150 μg/mL
Compound: Ciprofloxacin
|
Inhibition of metabolic activity in MG63 cells assessed as MTT reduction after 48 hrs
Inhibition of metabolic activity in MG63 cells assessed as MTT reduction after 48 hrs
|
[PMID: 17088489]
|
|
MG-63
|
IC50 |
160 μg/mL
Compound: Ciprofloxacin
|
Antiproliferative effect against MG63 cells assessed as BrdU incorporation into DNA after 48 hrs after 48 hrs
Antiproliferative effect against MG63 cells assessed as BrdU incorporation into DNA after 48 hrs after 48 hrs
|
[PMID: 17088489]
|
|
MG-63
|
IC50 |
|
Inhibition of human MG63 cell proliferation assessed as bromodeoxyuridine incorporation during DNA synthesis after 48 hrs by ELISA
Inhibition of human MG63 cell proliferation assessed as bromodeoxyuridine incorporation during DNA synthesis after 48 hrs by ELISA
|
[PMID: 19595598]
|
|
MRC5
|
IC50 |
181 μM
Compound: Ciprofloxacin
|
Cytotoxicity against human MRC5 cells
Cytotoxicity against human MRC5 cells
|
[PMID: 24206766]
|
|
MRC5
|
IC50 |
|
Cytotoxicity against human MRC5 cells assessed as reduction in cell viability incubated for 48 hrs by MTT assay
Cytotoxicity against human MRC5 cells assessed as reduction in cell viability incubated for 48 hrs by MTT assay
|
[PMID: 31431360]
|
|
MT4
|
CC50 |
60 μM
Compound: Ciprofloxacin
|
Concentration required to reduce the viability of mock-infected MT-4 cells by 50%
Concentration required to reduce the viability of mock-infected MT-4 cells by 50%
|
[PMID: 10397494]
|
|
NCI-H460
|
IC50 |
|
Inhibition of human NCI-H460 cell proliferation by sulphorodhamine B assay
Inhibition of human NCI-H460 cell proliferation by sulphorodhamine B assay
|
[PMID: 19595598]
|
|
NIH3T3
|
IC50 |
305.77 μg/mL
Compound: Ciprofloxacin
|
Cytotoxicity against mouse NIH/3T3 cells assessed as reduction in cell proliferation after 24 hrs by MTT assay
Cytotoxicity against mouse NIH/3T3 cells assessed as reduction in cell proliferation after 24 hrs by MTT assay
|
[PMID: 28174104]
|
|
NIH3T3
|
IC50 |
|
Inhibition of mouse NIH/3T3 cell proliferation by MTT assay
Inhibition of mouse NIH/3T3 cell proliferation by MTT assay
|
[PMID: 19595598]
|
|
NIH3T3
|
IC50 |
> 200 μM
Compound: Ciprofloxacin
|
Cytotoxicity against mouse NIH/3T3 cells after 48 hrs by SRB assay
Cytotoxicity against mouse NIH/3T3 cells after 48 hrs by SRB assay
|
[PMID: 23711920]
|
|
Osteoblast
|
IC50 |
170 μg/mL
Compound: Ciprofloxacin
|
Antiproliferative effect against primary human osteoblasts assessed as BrdU incorporation into DNA after 48 hrs
Antiproliferative effect against primary human osteoblasts assessed as BrdU incorporation into DNA after 48 hrs
|
[PMID: 17088489]
|
|
Osteoblast
|
IC50 |
> 400 μg/mL
Compound: Ciprofloxacin
|
Inhibition of metabolic activity in primary human osteoblasts assessed as MTT reduction after 48 hrs
Inhibition of metabolic activity in primary human osteoblasts assessed as MTT reduction after 48 hrs
|
[PMID: 17088489]
|
|
PBMC
|
IC50 |
> 0.5 mM
Compound: CPX, B-H
|
Cytotoxicity against human PBMC measured after overnight incubation by MTT assay
Cytotoxicity against human PBMC measured after overnight incubation by MTT assay
|
[PMID: 21855181]
|
|
PC-3
|
IC50 |
|
Antiproliferative activity against human PC3 cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
Antiproliferative activity against human PC3 cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
|
[PMID: 31678743]
|
|
PC-3
|
IC50 |
|
Antitumor activity against human PC3 cells after 5 days by MTT assay
Antitumor activity against human PC3 cells after 5 days by MTT assay
|
[PMID: 19595598]
|
|
SH-SY5Y
|
IC50 |
> 0.5 mM
Compound: CPX, B-H
|
Cytotoxicity against human SH-SY5Y cells measured after overnight incubation by MTT assay
Cytotoxicity against human SH-SY5Y cells measured after overnight incubation by MTT assay
|
[PMID: 21855181]
|
|
SK-MEL
|
IC50 |
|
Inhibition of human SK-MEL cell proliferation by MTT assay
Inhibition of human SK-MEL cell proliferation by MTT assay
|
[PMID: 19595598]
|
|
SK-MEL3
|
IC50 |
137 μM
Compound: Ciprofloxacin
|
Cytotoxicity against human SK-MEL-3 cells assessed as reduction in cell viability by MTT assay
Cytotoxicity against human SK-MEL-3 cells assessed as reduction in cell viability by MTT assay
|
[PMID: 30660827]
|
|
SK-MEL3
|
IC50 |
|
Anticancer activity against human SK-MEL3 cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
Anticancer activity against human SK-MEL3 cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
|
[PMID: 31881454]
|
|
SMMC-7721
|
IC50 |
137 μM
Compound: Ciprofloxacin
|
Cytotoxicity against human SMMC7721 cells assessed as reduction in cell viability by MTT assay
Cytotoxicity against human SMMC7721 cells assessed as reduction in cell viability by MTT assay
|
[PMID: 30660827]
|
|
SMMC-7721
|
IC50 |
>= 100 μM
Compound: Ciprofloxacin
|
Antiproliferative activity against human SMMC7721 cells by MTT assay
Antiproliferative activity against human SMMC7721 cells by MTT assay
|
[PMID: 30660827]
|
|
SW-620
|
IC50 |
|
Antiproliferative activity against human SW620 cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
Antiproliferative activity against human SW620 cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
|
[PMID: 31678743]
|
|
SW480
|
IC50 |
|
Inhibition of human SW480 cell proliferation by MTT assay
Inhibition of human SW480 cell proliferation by MTT assay
|
[PMID: 19595598]
|
|
SW480
|
IC50 |
137 μM
Compound: Ciprofloxacin
|
Cytotoxicity against human SW480 cells assessed as reduction in cell viability by MTT assay
Cytotoxicity against human SW480 cells assessed as reduction in cell viability by MTT assay
|
[PMID: 30660827]
|
|
SW480
|
IC50 |
|
Antiproliferative activity against human SW480 cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
Antiproliferative activity against human SW480 cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
|
[PMID: 31678743]
|
|
SW480
|
IC50 |
|
Anticancer activity against human SW480 cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
Anticancer activity against human SW480 cells assessed as reduction in cell viability incubated for 72 hrs by MTT assay
|
[PMID: 31881454]
|
|
Splenocyte
|
IC50 |
|
Cytotoxicity against concanavalin-stimulated BALB/c mouse splenocytes after 72 hrs by resazurin dye reduction method
Cytotoxicity against concanavalin-stimulated BALB/c mouse splenocytes after 72 hrs by resazurin dye reduction method
|
[PMID: 19908867]
|
|
THP-1
|
IC50 |
60.5 μM
Compound: Ciprofloxacin
|
Cytotoxicity against human THP1 cells assessed as cell viability after 72 hrs by MTT assay
Cytotoxicity against human THP1 cells assessed as cell viability after 72 hrs by MTT assay
|
[PMID: 24650715]
|
|
U-373MG ATCC
|
IC50 |
|
Antitumor activity against human U373MG cells after 5 days by MTT assay
Antitumor activity against human U373MG cells after 5 days by MTT assay
|
[PMID: 19595598]
|
|
V79
|
IC50 |
|
Mammalian cell cytotoxicity test in chinese hamster V79 cells (clonogenic cytotoxicity)
Mammalian cell cytotoxicity test in chinese hamster V79 cells (clonogenic cytotoxicity)
|
[PMID: 1469702]
|
|
V79
|
IC50 |
380 μg/mL
Compound: 4, Ciprofloxacin
|
Mammalian cell cytotoxicity test in chinese hamster V79 cells (clonogenic cytotoxicity)
Mammalian cell cytotoxicity test in chinese hamster V79 cells (clonogenic cytotoxicity)
|
[PMID: 1469702]
|
|
V79
|
IC50 |
|
Mammalian cell cytotoxicity test in chinese hamster V79 cells (clonogenic cytotoxicity)
Mammalian cell cytotoxicity test in chinese hamster V79 cells (clonogenic cytotoxicity)
|
[PMID: 1469702]
|
|
V79
|
IC50 |
|
Inhibition of chinese hamster V79 cell proliferation assessed as bromodeoxyuridine incorporation during DNA synthesis after 48 hrs by ELISA
Inhibition of chinese hamster V79 cell proliferation assessed as bromodeoxyuridine incorporation during DNA synthesis after 48 hrs by ELISA
|
[PMID: 19595598]
|
|
V79
|
IC50 |
> 200 μg/mL
Compound: 21 Ciprofloxacin
|
Clonogenic cytotoxicity against Hamster V-79 cells
Clonogenic cytotoxicity against Hamster V-79 cells
|
[PMID: 8145222]
|
|
V79
|
IC50 |
|
Clonogenic cytotoxicity against Chinese hamster V-79 cells
Clonogenic cytotoxicity against Chinese hamster V-79 cells
|
[PMID: 7473575]
|
|
V79-4
|
IC50 |
58.55 μg/mL
Compound: Ciprofloxacin (CPFX)
|
Cytotoxicity was determined for the compound in V79-4 cell line
Cytotoxicity was determined for the compound in V79-4 cell line
|
[PMID: 14980680]
|
|
Vero
|
CC50 |
|
Cytotoxicity against African green monkey Vero cells assessed as reduction in cell viability after 72 hrs by MTT assay
Cytotoxicity against African green monkey Vero cells assessed as reduction in cell viability after 72 hrs by MTT assay
|
[PMID: 30025351]
|
|
Vero
|
CC50 |
128 μg/mL
Compound: Ciprofloxacin
|
Cytotoxicity in African green monkey Vero cells
Cytotoxicity in African green monkey Vero cells
|
[PMID: 31358466]
|
|
Vero
|
CC50 |
|
Cytotoxicity against African green monkey Vero cells assessed as reduction in cell viability after 72 hrs by MTT assay
Cytotoxicity against African green monkey Vero cells assessed as reduction in cell viability after 72 hrs by MTT assay
|
[PMID: 30690301]
|
|
Vero
|
CC50 |
|
Cytotoxicity against african green monkey Vero cells after 72 hrs by MTT assat
Cytotoxicity against african green monkey Vero cells after 72 hrs by MTT assat
|
[PMID: 21146257]
|
|
Vero
|
IC50 |
192 μM
Compound: Ciprofloxacin
|
Cytotoxicity against african green monkey Vero cells
Cytotoxicity against african green monkey Vero cells
|
[PMID: 24206766]
|
|
Vero
|
IC50 |
> 188.5 μM
Compound: Ciprofloxacin
|
Cytotoxicity against african green monkey Vero cells after 72 hrs by MTT assay
Cytotoxicity against african green monkey Vero cells after 72 hrs by MTT assay
|
[PMID: 19131245]
|
|
WI-38
|
IC50 |
> 100 μM
Compound: Ciprofloxacin
|
Antiproliferative activity against human WI38 cells after 24 hrs by BrdU incorporation assay
Antiproliferative activity against human WI38 cells after 24 hrs by BrdU incorporation assay
|
[PMID: 27555286]
|